CBD’s Moment Meets Regulation: Reformulating for a Safer Future
CBD has moved from novelty to necessity in wellness aisles across the UK. Once found only in niche health stores, cannabidiol has now become a mainstay in the mainstream — embraced by consumers seeking support for everything from anxiety and stress relief to sleep improvement and general wellness.
Today, UK shelves are lined with an expanding array of CBD-infused products: gummies, oils, tinctures, capsules, functional beverages, chocolates, and more. The diversity is exciting — but also increasingly scrutinized.
As consumer interest has surged, so too has the attention of regulators. On 1 July 2025, the UK Food Standards Agency (FSA) introduced a significant revision to its CBD safety guidance for food and supplement products. More than 12,000 CBD products currently listed on the FSA’s public register must now reassess their formulations in light of the updated rules.
This is not just another compliance box to check. It’s a defining moment for the CBD industry in the UK — one where scientific rigor, product innovation, and strategic compliance must converge.

The Science and Scrutiny behind CBD
CBD (cannabidiol) is a non-psychoactive compound found in hemp and cannabis plants. It interacts with the body’s endocannabinoid system (ECS), a regulatory network involved in maintaining homeostasis, modulating the immune system, and regulating neurotransmitter signaling.
Research indicates that CBD may offer several potential health benefits. Studies suggest it could help reduce anxiety and stress, improve sleep quality, and alleviate chronic pain. Additionally, CBD has been associated with anti-inflammatory effects and may play a role in managing symptoms of certain neurological conditions, such as epilepsy.
As this natural compound has gained global recognition, its inclusion in foods, supplements, and wellness products has increased significantly. But increasing exposure demands clear safety thresholds, accurate labeling, and scientific justification — which is precisely what the FSA’s latest update addresses.
What Prompted the New CBD Limits
The updated FSA guidance is based on a growing body of toxicological and clinical data. Key concerns include:
- Liver toxicity: Elevated liver enzymes in animal and human studies point to potential hepatotoxicity at high doses.
- Reproductive and developmental toxicity: Animal studies suggest possible impacts on fertility and fetal development.
- Drug interactions: CBD may inhibit liver enzymes that metabolize certain drugs, potentially leading to harmful interactions with standard medications.
- Insufficient long-term safety data: Despite years of consumer use, there remains a lack of long-term clinical studies on chronic daily CBD consumption.
The new guidance reflects a precautionary approach, prioritizing consumer safety over commercial momentum.
It also brings the UK’s standards closer to international benchmarks set by the European Food Safety Authority (EFSA) and the U.S. Food and Drug Administration (FDA), while maintaining the FSA’s commitment to oversight of novel foods.
Significant Change, Small Window: What the New FSA Guidance Means
The guidance affects all businesses with products on the FSA’s Public List and introduces:
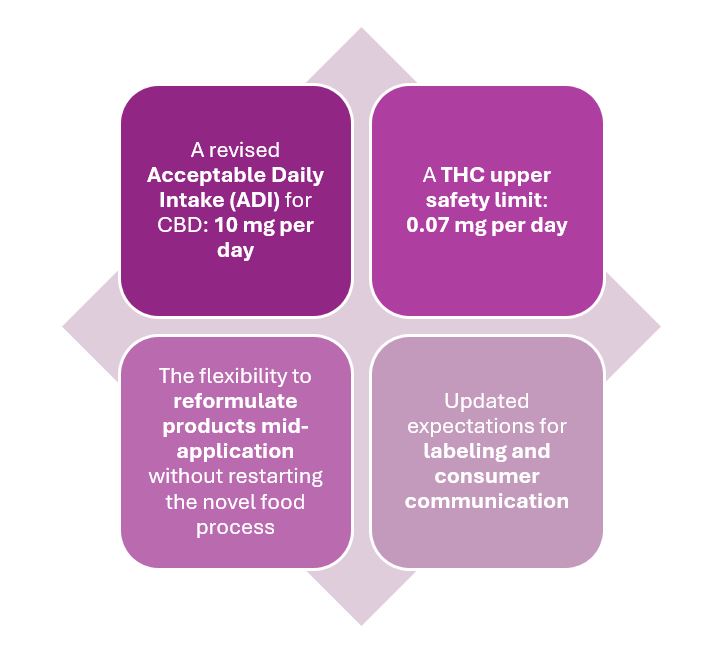
These changes are not just procedural — they signal a broader shift toward evidence-based risk management in the UK CBD space.
What This Means for CBD Businesses
Now is the time for CBD companies to take a hard look at their product portfolios and regulatory strategies. Questions every brand should be asking include:
- Does each product align with the new FSA safety thresholds?
- Have I updated my toxicological assessments and ADI derivations?
- Are my exposure models and novel food dossiers up to date?
- Do my labels reflect current consumer safety expectations?
Meeting these standards isn’t just about avoiding regulatory action; it’s also about ensuring compliance. It’s about embracing a science-led, safety-first approach that builds credibility with both regulators and consumers.
The good news? Reformulation doesn’t mean starting from scratch. It means evolving — responsibly and strategically — to build products that are both innovative and compliant.
Evalueserve’s Expertise: From Safety Science to Strategic Execution
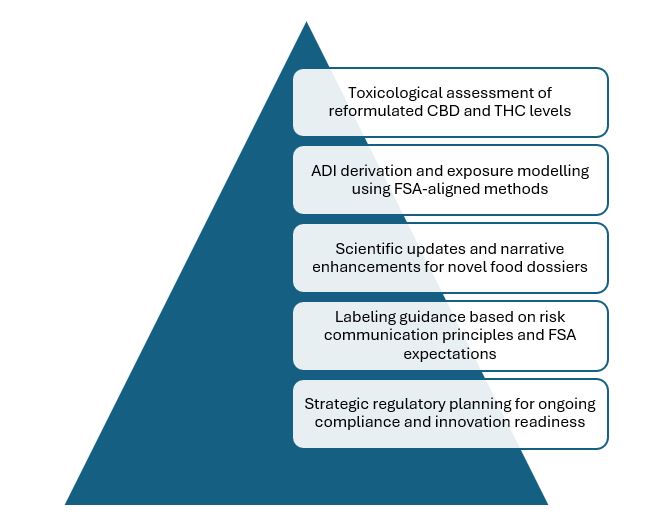
At Evalueserve IP and R&D, we combine toxicological rigor, regulatory insight, and market alignment to help clients adapt swiftly and confidently to regulatory change. Our toxicology consulting services include:
Why Work with Evalueserve?
Whether you’re refining an existing formulation or evaluating a new product, we ensure your decisions are data-driven and fully aligned with UK regulatory frameworks.
At Evalueserve, we blend toxicological rigor, regulatory insight, and market awareness to help clients adapt swiftly and confidently to evolving requirements. Our team supports projects ranging from toxicological assessments of reformulated CBD and THC levels to ADI derivation, exposure modelling, and strategic planning for compliance and innovation readiness.
Let’s Get You Compliant, Fast
We understand the urgency. Whether you’re proactively reformulating or seeking to justify existing levels, we can support you from science to strategy — fast. The FSA’s updated guidance offers a clear signal: product safety and regulatory alignment will define long-term success in the CBD category. Reformulation, when done strategically, isn’t just a compliance task — it’s a brand opportunity.
Evalueserve is here to support every step of the journey — from technical assessments to regulatory submissions and beyond.
Talk to One of Our Experts
Connect with us today to schedule a reformulation strategy session or dossier review.
Let’s build safer, more innovative CBD products — together.



